 Image credit: ACS
Image credit: ACSAbstract
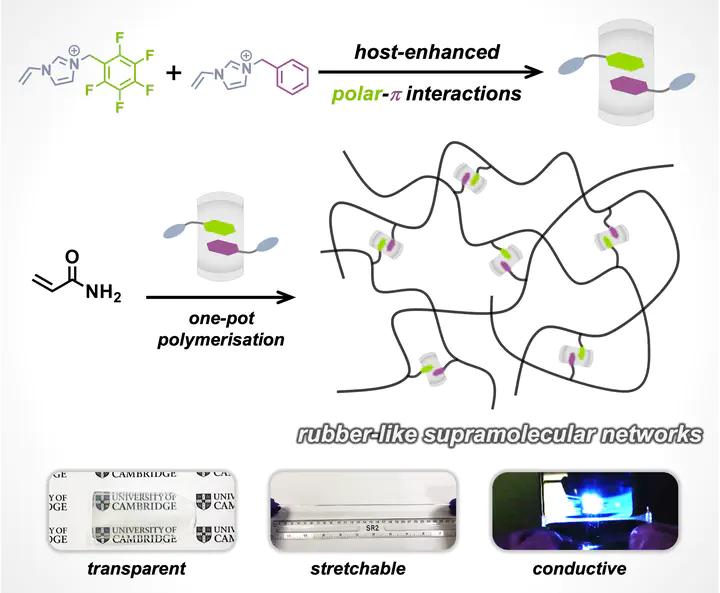
Phenyl-perfluorophenyl polar−π interactions have been revisited for the design and fabrication of functional supramolecular systems. The relatively weak associative interactions (ΔG ≈ −1.0 kcal/mol) have limited their use in aqueous self-assembly to date. Herein, we propose a strategy to strengthen phenyl-perfluorophenyl polar−π interactions by encapsulation within a synthetic host, thus increasing the binding affinity to ΔG = −15.5 kcal/mol upon formation of heteroternary complexes through social self-sorting. These heteroternary complexes were used as dynamic, yet strong, cross-linkers in the fabrication of supramolecular gels, which exhibited excellent viscoelasticity, stretchability, self-recovery, self-healing, and energy dissipation. This work unveils a general approach to exploit host-enhanced polar−π interactions in the design of robust aqueous supramolecular systems.